|
(Remember that the convention for writing formulas for ionic compounds is not to include the ionic charge.) This is a clue that the other part of the formula, Ba, is actually the Ba 2+ ion, with the 2+ charge balancing the overall 2− charge from the two nitrate ions. For example, if you see the formula Ba(NO 3) 2, you may recognize the “NO 3” part as the nitrate ion, NO 3 −. Second, if you recognize the formula of a polyatomic ion in a compound, the compound is ionic. Also note that this combination of nitrogen and oxygen has no electric charge specified, so it is not the nitrite ion. Oxygen has six valence electrons, which are the electrons in the outer shell of the electron. It is not an ionic compound it belongs to the category of covalent compounds discussed elsewhere. The ion name for oxygen is oxide.The charge of an oxygen ion is -2. Oxygen is in group 6 of the periodic table. When atoms gain electron/s, the negatively charged ion is formed, and when the atoms lose electron/s, the positively charged ion is formed. This electric charge generated on the ion is known as Ionic charge. (In fact, it is ionic.) In contrast, the compound NO 2 contains two elements that are both nonmetals (nitrogen, from group 15 (or 5A), and oxygen, from group 16 (or 6A). For elements in groups 6 and 7, the charge on the ion relates to the group number of the element in the periodic table. Ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). Therefore, it is most likely an ionic compound. For example, CaBr 2 contains a metallic element (calcium, a group 2 (or 2A) metal) and a nonmetallic element (bromine, a group 17 (or 7A) nonmetal). First, compounds between metal and nonmetal elements are usually ionic. There are two ways to recognize ionic compounds. 
This is to show that the subscript applies to the entire polyatomic ion. If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. Unfortunately, the relative nature of these suffixes mandates that the ion formula/ion name combinations of the polyatomic ions must simply be memorized.\): Some Polyatomic Ions Name Additionally, both the nitrate ion and the sulfite ion contain three oxygens, but these polyatomic ions do not share a common suffix. While both the nitrate ion and the sulfate ion share an " -ate" suffix, the former contains three oxygens, but the latter contains four. 
However, the sulfate ion is symbolized as SO 4 −2. For example, the nitrate ion, which is symbolized as NO 3 −1, has one more oxygen than the nitrite ion, which is symbolized as NO 2 −1. With Al 3 + and O 2, note that neither charge is a perfect multiple of the other. 
We need three F ions to balance the charge on the Al 3 + ion, so the proper ionic formula is AlF 3. Note that all of the polyatomic ions whose names end in " -ate" contain one more oxygen than those polyatomic anions whose names end in " -ite." Unfortunately, these suffixes only indicate the relative number of oxygens that are contained within the polyatomic ions. We need two Cl ions to balance the charge on one Ca 2 + ion, so the proper ionic formula is CaCl 2. 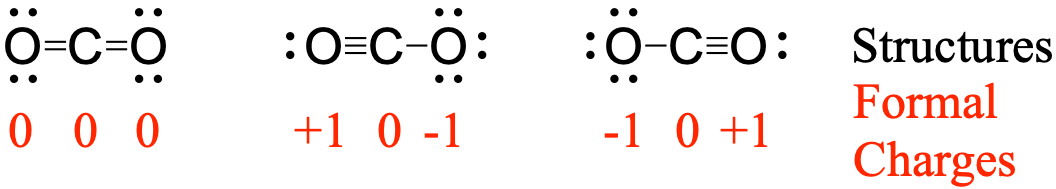
Additionally, these suffixes also indicate the relative number of oxygens that are contained within the polyatomic ions. Lithium-ion batteries, while commonplace in today’s world powering everything from electric vehicles to smartphones. Therefore, " -ate" and " -ite" suffixes are employed, in order to denote that the corresponding polyatomic ions are part of a series. Researchers at TU Wien (Vienna) have developed a groundbreaking oxygen-ion battery, which boasts exceptional durability, eliminates the need for rare elements, and solves the problem of fire hazards. A single suffix, "-ide," is insufficient for distinguishing the names of the anions in a related polyatomic series. The remaining polyatomic anions, which all contain oxygen, in combination with another non-metal, exist as part of a series in which the number of oxygens within the polyatomic unit can vary. However, only two of these, the hydroxide ion and the cyanide ion, are named using the "-ide" suffix that is typically indicative of negatively-charged particles. The remaining polyatomic ions are all negatively-charged and, therefore, are classified as anions. This polyatomic ion contains one nitrogen and four hydrogens that collectively bear a +1 charge. Note that only one polyatomic ion in this Table, the ammonium ion (NH 4 +), is a cation. \): Ion Names and Ion Formulas of Common Polyatomic Ions Ion Name
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
